Medications List
Medications List
Source PDF: medlistqig.pdf
Quick Implementation Guide
Edition 1 20 [th] May 2010
Copyright ©2013 Health and Social Care Information Centre
This document was prepared for NHS Connecting for Health which ceased to exist on 31 March 2013. It may contain references to organisations, projects and other initiatives which also no longer exist. If you have any questions relating to any such references, or to any other aspect of the content, please contact cuistakeholder.mailbox@hscic.gov.uk
Copyright: You may re-use this information (excluding logos) free of charge in any format or medium, under the terms of the Open Government Licence. To view this licence, visit nationalarchives.gov.uk/doc/open-governmentlicence or email psi@nationalarchives.gsi.gov.uk.
1
Copyright ©2013 Health and Social Care Information Centre
Guidance
Introduction
An electronic display of a patient’s medications must be able to support the complex information needs of a wide range of health care professions and settings. A successful display solution must therefore take those information needs into account whilst ensuring patient safety and promoting consistency across views and between systems.
A list of medications is expected to be one of many ways of displaying medications information for a single patient. The information required for a medication review will vary for different contexts so guidance is provided for the needs that are common to all kinds of medications review. This list of medications forms only part of the Medications Management views and tools that would be available in a clinical application.
Important
Although it contains all of the guidance points, this document omits supporting information and is therefore not intended as a substitute for the full NHS CUI documents Medications List [1] . Refer to that document for the definitive statement of the design guidance in this area.
The visual representations used to display the guidance are illustrative only. Stylistic choices (for example, display font used), unless otherwise specified, are not part of the guidance and are therefore not mandatory requirements.
1 Medications Management – Medications List – User Interface Design Guidance (v1.0.0.0)
2
Copyright ©2013 Health and Social Care Information Centre
Guidance
Structure and Layout
This simplified diagram shows some of the elements of a Medications List View:
Current and Past Menu Buttons
Column Headings
Medications
Group Headings
Notification
Important


Look-ahead Scroll Bar (LASB) Notification
Look-ahead Scroll Bar Notification
| Col1 | Drug Details | Col3 | yyyy | zzzz | Start ▼ | Col7 | Col8 |
|---|---|---|---|---|---|---|---|
| ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz aaa·** bbb** 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** kkk– xxxx - xxxx yyyy zzzz Group A(2) Group B 3 past medications were completed… | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz aaa·** bbb** 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** kkk– xxxx - xxxx yyyy zzzz Group A(2) Group B 3 past medications were completed… | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz aaa·** bbb** 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** kkk– xxxx - xxxx yyyy zzzz Group A(2) Group B 3 past medications were completed… | aaa· | aaa· | ** bbb** | ** bbb** | |
| ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz aaa·** bbb** 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** kkk– xxxx - xxxx yyyy zzzz Group A(2) Group B 3 past medications were completed… | 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** | 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** | 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** | 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** | 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** | 2 more·** kkk**·** jjj**·** iii · ggg**·** fff** |
For the sake of brevity, illustrations in sections other than the Look-Ahead Scroll Bar section omit LASB notifications. Refer to that section for specific guidance on reserving space for, and displaying, LASB notifications.
3
Copyright ©2013 Health and Social Care Information Centre
Structure and Layout
Tabular Layout
Guidance
MEDv-020
Present medications as lines of text within rows in a tabular format where each row represents one medication
Mandatory

MEDv-141
Use composite columns to minimise the display of blank cells for some rows (that is, avoid placing each individual data point in a separate column)
Recommended
4
Copyright ©2013 Health and Social Care Information Centre
composite column
Guidance
Structure and Layout
Indicating List Length
| Drug Details | Status | Start Date ▼ |
|---|---|---|
| 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required |
5
MEDv-142
When the list is scrolled to the end, display a space at the bottom of the list with a height equivalent to a line of text
Recommended
Copyright ©2013 Health and Social Care Information Centre
Structure and Layout
Gridlines and Row Formatting
| temazepam – tablet – DOSE 20 mg – oral – at night Started 26-May-2010 | Col2 |
|---|---|
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required |
MEDv-021
Avoid the use of strong grids and strong vertical lines. (Use subtle methods to support distinguishing between rows in the list)
Mandatory
MEDv-022
Use subtle alternate row shading
Recommended
MEDv-143
Use at least alternate row shading or lines between rows
Mandatory
When using alternate row shading, ensure that colour and brightness of the background does not interfere with the readability of the foreground text
Mandatory
MEDv-145
Supplement alternate shading with 1 point pale lines between rows
Recommended
6
Copyright ©2013 Health and Social Care Information Centre
Guidance
Subtle gridlines between rows
MEDv-144
Guidance
Structure and Layout
Displaying Icons for Drug Details
| Drug Details column | Col2 |
|---|---|
| 28-May-2010 B glyceryl trinitrate – 400 micrograms per dose – sublingual spray – DOSE 1 to 2 puffs– sublingual –as required – maximum 3 doses in 15 minutes Started | 28-May-2010 B glyceryl trinitrate – 400 micrograms per dose – sublingual spray – DOSE 1 to 2 puffs– sublingual –as required – maximum 3 doses in 15 minutes Started |
| 27-May-2010 Started B A lansoprazole – orodispersible tablet – DOSE 30 mg– oral – once a day | 27-May-2010 Started B A lansoprazole – orodispersible tablet – DOSE 30 mg– oral – once a day |
| 27-May-2010 Started A dalteparin – 10,000 units per 1 mL – injection – DOSE 10,000 units– subcutaneous – twice a day | 27-May-2010 Started A dalteparin – 10,000 units per 1 mL – injection – DOSE 10,000 units– subcutaneous – twice a day |
MEDv-023
Support the display of icons following the text of the Drug Details column in the Medications List View
Mandatory
Note The lettered squares that have been used to represent icons are placeholders only and do not form part of the guidance
Copyright ©2013 Health and Social Care Information Centre
icon
7
Guidance
Structure and Layout
Empty Lists
| Drug Details | Status | First Administration |
|---|---|---|
| There are no current medications for this patient | There are no current medications for this patient | There are no current medications for this patient |
MEDv-146
Display a message when a list is empty (for example, when there are no current medications)
Mandatory
8
Copyright ©2013 Health and Social Care Information Centre
Guidance
Columns
Mandatory Columns
| drug details status information an initiation date | Col2 | Col3 |
|---|---|---|
| Drug Details | Status | Start Date ▼ |
| 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
MEDv-147
Provide a column that contains status information including information that defines whether the medication is ‘current’ or ‘past’
Mandatory
MEDv-148
Provide a column that contains drug details according to Medication Line [1] guidance
Mandatory
1 Medications Management – Medication Line – User Interface Design Guidance (v2.0.0.0)
9
Copyright ©2013 Health and Social Care Information Centre
Columns
Mandatory Columns
| Drug Details | Status | Start Date ▼ |
|---|---|---|
| 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous –with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous –with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous –with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral –once a day 24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral –once a day 24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral –once a day 24-May-2010 Started |
MEDv-149
Guidance
When displaying current medications, provide a column that contains an initiation date (such as the date of the first planned administration). The examples in this document show a Start Date column
Mandatory
| Status | Drug Details | End Date ▲ |
|---|---|---|
hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 | hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 | hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 |
methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 | methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 | methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 |
furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 | furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 | furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 |
MEDv-150
When displaying past medications, provide a column that contains a stop date (such as the date of the last administration or the date that the medication was discontinued). The examples in this document show an End Date column
Mandatory
10
Copyright ©2013 Health and Social Care Information Centre
Guidance
Columns
Date Columns
The specific information to display in date columns is not part of guidance because it needs to differ between tasks and contexts. Guidance is therefore provided for the relative placement of date columns.
Illustrations in this document use column headings with the following labels:
-
Start Date for the start or initiation of a medication
-
End Date for the end or completion of a medication
| Drug Details | Status | Col3 | Start Date ▼ | End Date |
|---|---|---|---|---|
| 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 |
MEDv-154
Maintain consistent placement of date columns relative to one another and relative to the Drug Details column in both current and past medications
Mandatory
11
When an end date column is displayed, place a start date column before (to the left of) the end date column
Mandatory
MEDv-152
When an end date column is displayed, and there is no duration column, place a start date column adjacent to the end date column
Recommended
Copyright ©2013 Health and Social Care Information Centre
MEDv-151
Columns
Date Columns
Guidance
fixed width fixed width
| Drug Details | Status | Col3 | Start Date ▼ | End Date |
|---|---|---|---|---|
| 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 |
MEDv-153
Use fixed width columns for dates
Mandatory
12
Copyright ©2013 Health and Social Care Information Centre
Guidance
Columns
Column Headings
Drug Details Status Start Date ▼ End Date
MEDv-155
Label columns with text that describes the contents unambiguously and succinctly (such as, ‘Status’, ‘Date Prescribed’ or ‘First Administration’)
Recommended
MEDv-156
Use a unique heading for each column
Mandatory
13
Copyright ©2013 Health and Social Care Information Centre
Columns
Composite Columns
Review and End Dates ▼
Review 06-Jun-2010 End 12-Jun-2010
Review 08-Jun-2010 End 08-Jun-2010
Guidance
MEDv-027
Allow columns to contain more than one attribute for a single medication
Mandatory
MEDv-157
When combining two attributes that have the same data type (such as dates), include labels for both attributes in the column heading
Mandatory
MEDv-158
When combining two attributes that have the same data types (such as dates), include labels for both attributes within the cell
Mandatory
Copyright ©2013 Health and Social Care Information Centre
14
Guidance
Columns
Constraining Dimensions
MEDv-159
Maintain the relative proportions of columns such that the Drug Details column is the widest
Mandatory
MEDv-161
Define minimum widths for all columns
Recommended
| widest column | Col2 | Col3 | Col4 | Col5 |
|---|---|---|---|---|
| Drug Details | Status | Start Date ▼ | End Date | |
| 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night03-Jun-2010 |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag 09-Jun-2010 |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started 06-Jun-2010 |
15
limited number of columns
MEDv-160
Avoid the need for horizontal scrolling by limiting the number of columns visible at any one time
Mandatory
Copyright ©2013 Health and Social Care Information Centre
Status
Guidance
MEDv-162
Ensure that all medications have a status value and the status cannot be blank
Mandatory
MEDv-163
Limit status descriptions to short phrases, preferably no more than two words
Recommended
MEDv-042
Display the status of each medication in bold
Mandatory
MEDv-164
Allow status to be supplemented with additional information (such as pharmacy verified)
Recommended
MEDv-165
Use the status description to differentiate between medications that have no recorded administration events and those that have
Mandatory
| Col1 | Col2 | Col3 | Col4 |
|---|---|---|---|
| Status | Star | ||
| Not Started 29- | Not Started 29- | Not Started 29- | M M M |
| Started 25- | Started 25- | Started 25- | Started 25- |
| Started 24- | Started 24- | Started 24- | Started 24- |
| Col1 | Col2 | Col3 | Col4 |
|---|---|---|---|
| Status | Star | ||
| Not Started Verified 29- | Not Started Verified 29- | Not Started Verified 29- | M |
| Started Not Verified 25- | Started Not Verified 25- | Started Not Verified 25- | M |
| Started Verified 24- | Started Verified 24- | Started Verified 24- | M |
Copyright ©2013 Health and Social Care Information Centre
16
Guidance
Status
17
MEDv-167
Assign a status of ‘Started’ to medications that have an administration event recorded and have further scheduled administrations
Recommended
MEDv-168
Assign a status of ‘Not Started’ to medications that have an administration scheduled and a start date in the future
Recommended
MEDv-170
Assign a status of ‘Completed’ to medications that have administration events recorded according to their schedule (within tolerances) and have an end date in the past
Recommended
MEDv-171
Assign a status of ‘Discontinued’ to medications that were stopped on a date that preceded one or more of the scheduled administrations
Recommended
Copyright ©2013 Health and Social Care Information Centre
| Col1 | Col2 | Col3 | Col4 |
|---|---|---|---|
| Status | Star | ||
| Started 25- | Started 25- | Started 25- | M |
| Col1 | Col2 | Col3 | Col4 |
|---|---|---|---|
| Status | Star | ||
| Not Started 29- | Not Started 29- | Not Started 29- | M |
| Status | Drug Details | Col3 |
|---|---|---|
| Completed hydrochl | Completed hydrochl |
| Status | Drug Details | Col3 |
|---|---|---|
| Discontinued methyld | Discontinued methyld |
Status
Guidance
MEDv-166
Support a status of ‘suspended’ and include medications with this status in current medications
Mandatory
MEDv-169
Assign a status of ‘Suspended’ to medications that are marked as not to be administered but which are intended to be resumed at a later date
Recommended
MEDv-024
Use visual design to draw attention to suspended medications
Mandatory
| Col1 | Status | Star | Col4 |
|---|---|---|---|
| Suspended on 30-May-2008 | 24- | M |
Copyright ©2013 Health and Social Care Information Centre
18
Guidance
Status
| Drug Details | Status | Start Date ▼ |
|---|---|---|
| 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous –with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous –with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous –with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral –once a day 24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral –once a day 24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral –once a day 24-May-2010 Started |
MEDv-025 MEDv-172
Use visual design to distinguish a list of current medications from a list of past medications
MEDv-025
Mandatory
Define medications with a status of either ‘Started’, ‘Not Started’ or ‘Suspended’ as current medications
Recommended
| Status | Drug Details | End Date ▲ |
|---|---|---|
hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 | hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 | hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 |
methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 | methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 | methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 |
furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 | furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 | furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 |
19
Define medications with a status of ‘Completed’ or ‘Discontinued’ as past medications
Recommended
Copyright ©2013 Health and Social Care Information Centre
MEDv-173
Current and Past Medications
Controls
| Current Past | Past | Col3 |
|---|---|---|
default selection
one is selected
Guidance
MEDv-063
Provide buttons for displaying current and past medications respectively in the Medications List View and label the buttons ‘Current’ and ‘Past’
Mandatory
MEDv-062
Present the Medications List View with Current selected by default
Mandatory
MEDv-065
Do not allow Current and Past buttons to be selected simultaneously
Mandatory
MEDv-174
Ensure that either the Current or the Past button is selected at any one time
Mandatory
Copyright ©2013 Health and Social Care Information Centre
20
Guidance
Current and Past Medications
Controls
Example of Current and Past buttons
Example of Current and Past buttons in an alternative style
MEDv-064
Use the visual formatting of the Current and Past buttons to indicate which is currently selected
Mandatory
MEDv-066
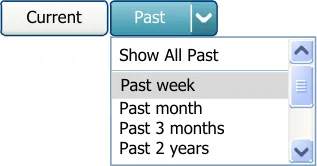
Supplement the Past button in the Medications List View with a drop-down control for displaying, selecting and applying a filter on the past medications view
Mandatory
MEDv-067
Include an option for displaying all past medications in the drop-down control
Mandatory
Copyright ©2013 Health and Social Care Information Centre
21

Current and Past Medications
Current Medications
| Drug Details | Status | Start Date ▼ |
|---|---|---|
| 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night | 26-May-2010 Started temazepam– tablet – DOSE 20 mg– oral – at night |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous –with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous –with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous –with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral –once a day 24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral –once a day 24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral –once a day 24-May-2010 Started |
MEDv-099
Guidance
By default, present current medications sorted reverse chronologically by a starting date, such that the most recent is first (top) in the list
Mandatory
MEDv-173
When displaying current medications, place the drug details in the first (furthest left) column
Mandatory
Copyright ©2013 Health and Social Care Information Centre
22
Guidance
Current and Past Medications
Recent Past Notifications
| Drug Details | Status | Start Date ▼ |
|---|---|---|
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required |
| 2 past medications were completed or discontinued in the last 48 hours | 2 past medications were completed or discontinued in the last 48 hours | 2 past medications were completed or discontinued in the last 48 hours |
MEDv-074
23
When displaying current medications, display a notification for medications that have been completed or discontinued within a specified time interval from the current time
Mandatory
MEDv-174
Use formatting to distinguish the recent past notifications from medications in the list
Recommended
Copyright ©2013 Health and Social Care Information Centre
Guidance
Current and Past Medications
Recent Past Notifications
Time interval
MEDv-075
Clearly display the time interval within the recent past notification
Mandatory
count
MEDv-077
Display a count of the number of recently past medications within the recent past notification in the medication list
Mandatory
Copyright ©2013 Health and Social Care Information Centre
24
Guidance
Current and Past Medications
Past Medications
First Column
Second Column
MEDv-175
When displaying past medications, place the status column first (furthest left) and the Drug Details column second
Mandatory
| Status Drug Details | Col2 | Col3 |
|---|---|---|
| Status | Drug Details | End Date ▲ |
hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 | hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 | hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 |
methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 | methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 | methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 |
furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 | furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 | furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 |
25
By default, sort medications reverse chronologically by end date (or equivalent) such that the most recent is first (top) when the filter is set to ‘Past’ in the Medications List View
Recommended
Copyright ©2013 Health and Social Care Information Centre
MEDv-100
Filtering
Filtering Past Medications
Guidance
MEDv-069
When a filter is applied to past medications in Medications List View, the Past button should indicate that it is currently selected
Mandatory
| Status | Drug Details | End Date ▲ |
|---|---|---|
| The total list (54) is filtered to show: Past 2 months (3) Remove Filter | The total list (54) is filtered to show: Past 2 months (3) Remove Filter | The total list (54) is filtered to show: Past 2 months (3) Remove Filter |
hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 | hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 | hydrochlorothiazide – tablet –DOSE 100 mg – oral – 4 times a dayCompleted 26-May-2010 |
methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 | methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 | methyldopa – tablet –DOSE 250 mg – oral – twice a dayDiscontinued 25-May-2010 |
furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 | furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 | furosemide – tablet –DOSE 40 mg – oral – once a dayCompleted 24-May-2010 |
MEDv-070
When a filter is applied to past medications in the Medications List View, display a filter notification at the top of the list below the column headings and above the scroll bar (thus ‘pushing’ the list of medications down a line)
Mandatory
Copyright ©2013 Health and Social Care Information Centre
26
Guidance
Filtering
Filtering Past Medications
MEDv-068
When a filter notification is displayed, include a control for removing the filter within that notification
Mandatory
MEDv-071
Display a description of the filter in use within the filter notification in the Medications List View
Mandatory
count of unfiltered total
count of filtered total
MEDv-072
Include a count of the number of medications displayed and a count of the total (unfiltered) number of past medications in a filter notification
Recommended
MEDv-176
Clearly label the counts (number of medications displayed and total unfiltered number) with text that allows them to be differentiated
Recommended
Copyright ©2013 Health and Social Care Information Centre
27
Guidance
Look-Ahead Scroll Bar
A look-ahead scroll bar (LASB) is a standard scroll bar that is supplemented with notifications at the top and bottom to indicate that there are items in the list that are not currently visible.

Look-ahead scroll bar notification
Look-ahead scroll bar
Look-ahead scroll bar notification
Medications
| Drug Details | Col2 | yyyy | zzzz | Start ▼ | Col6 |
|---|---|---|---|---|---|
| aaa· | aaa· | ** bbb** | |||
| ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz iii– xxxx - xxxx yyyy zzzz jjj– xxxx - xxxx yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz iii– xxxx - xxxx yyyy zzzz jjj– xxxx - xxxx yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz iii– xxxx - xxxx yyyy zzzz jjj– xxxx - xxxx yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz iii– xxxx - xxxx yyyy zzzz jjj– xxxx - xxxx yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz iii– xxxx - xxxx yyyy zzzz jjj– xxxx - xxxx yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz iii– xxxx - xxxx yyyy zzzz jjj– xxxx - xxxx yyyy zzzz |
| 2 more ·** ooo**·** nnn**·** mmm · lll**·** kkk** | 2 more ·** ooo**·** nnn**·** mmm · lll**·** kkk** | 2 more ·** ooo**·** nnn**·** mmm · lll**·** kkk** | 2 more ·** ooo**·** nnn**·** mmm · lll**·** kkk** | 2 more ·** ooo**·** nnn**·** mmm · lll**·** kkk** |
Alert Icon
Diagram of a Medications List View
MEDv-177
When displaying a list of current or past medications, and the scroll bar is active because the list is longer than the space available to display them, provide a clear indication that there are medications out of view
Mandatory
MEDv-178
When displaying current medications, supplement the standard scroll bar with notifications that display the names of drugs that are out of view. This document refers to this kind of scroll bar as a look-ahead scroll bar (LASB)
Recommended
Copyright ©2013 Health and Social Care Information Centre
28
Guidance
Look-Ahead Scroll Bar
Layout

MEDv-179
When displaying a LASB, reserve a space at the top and bottom of the list for look-ahead notifications
Mandatory
MEDv-180
Use a pale solid background colour for the space reserved for look-ahead notifications that is sufficient to distinguish the space from the background of the list
Recommended
Copyright ©2013 Health and Social Care Information Centre
| space | Col2 | Col3 | Col4 |
|---|---|---|---|
| space | space | space | space |
| Drug Details | yyyy | zzzz | Start ▼ |
| ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz fff– xxxx - xxxx yyyy zzzz ggg– xxxx - xxxx yyyy zzzz hhh– xxxx - xxxx yyyy zzzz |
| pale colour Status Start Date ▼ aspirin · paracetamol | Col2 | Col3 | Col4 |
|---|---|---|---|
| Status | Status | Start Date ▼ | Start Date ▼ |
| aspirin·** paracetamol** | aspirin·** paracetamol** | aspirin·** paracetamol** | |
29
Guidance
Look-Ahead Scroll Bar
Grouping
| Drug Details | Status | Col3 | Start Date ▼ |
|---|---|---|---|
| aspirin·** paracetamol** | aspirin·** paracetamol** | ||
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required Inhaled Rectal(1) Intravenous(3) | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required Inhaled Rectal(1) Intravenous(3) | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required Inhaled Rectal(1) Intravenous(3) | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled –four times a day as required Inhaled Rectal(1) Intravenous(3) |
MEDv-181
Example in which the first group in the list has been scrolled out of view. This group contains aspirin and paracetamol.

When grouping is applied, and there is a collapsed group out of view, display drug names in the look-ahead scroll bar for any drug that is out of view, irrespective of whether it is within a collapsed group or an expanded group
Recommended
MEDv-185
Do not include additional text or formatting to indicate grouping in the look-ahead notifications
Mandatory
Copyright ©2013 Health and Social Care Information Centre
30
Guidance
Look-Ahead Scroll Bar
Interaction
| Col1 | Col2 | Col3 |
|---|---|---|
| ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz | ddd– xxxx - xxxx eee– xxxx - xxxx ccc– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz |
MEDv-058
Update the look-ahead notifications dynamically in response to scrolling
Mandatory
MEDv-059
Allow the look-ahead notification to change width dynamically to accommodate its contents up to the available width
Mandatory
MEDv-182
Do not allow the look-ahead notification to be used for navigation by clicking on areas of the notification, such as drug names or counts
Mandatory
Copyright ©2013 Health and Social Care Information Centre
| Status Start Date ▼ aspirin · paracetamol | Col2 | Col3 | Col4 |
|---|---|---|---|
| Status | Status | Start Date ▼ | Start Date ▼ |
| aspirin·** paracetamol** | aspirin·** paracetamol** | aspirin·** paracetamol** | |
| Status Start Date ▼ aspirin · paracetamol | Col2 | Col3 | Col4 |
|---|---|---|---|
| Status | Status | Start Date ▼ | Start Date ▼ |
| aspirin·** paracetamol** | aspirin·** paracetamol** | aspirin·** paracetamol** | |
31
Look-Ahead Scroll Bar
aspirin - paracetamol
notification
up arrow
Guidance
MEDv-044
Restrict the look-ahead notifications to a single line each
Mandatory
MEDv-043
The look-ahead notifications should be clearly joined to the ‘up’ and ‘down’ arrow controls of the scroll bar respectively
Mandatory
MEDv-045
Do not place controls or other notifications such that they separate the look-ahead notification from the medications in the Medication List View
Mandatory
| Start Date ▼ aspirin · paracetamol | Col2 | Col3 |
|---|---|---|
| Start Date ▼ | ||
| aspirin·** paracetamol** | aspirin·** paracetamol** | |

notification
medications
Copyright ©2013 Health and Social Care Information Centre
32
Guidance
Look-Ahead Scroll Bar
Order of Contents

MEDv-049
The order of both the items in the look-ahead notification and the medications list should always be the same
Mandatory
MEDv-048
The look-ahead notification is populated from right to left such that the next drug in the list appears closest to the scroll bar
Mandatory
| Col1 | Col2 | 1 · 2 · 3 · 4 · 5 |
|---|---|---|
| **7 **– xxxx - xxxx **8 **– xxxx - xxxx **6 **– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz **9 **– xxxx - xxxx 10– xxxx - xxxx yyyy zzzz yyyy zzzz 11– xxxx - xxxx yyyy zzzz | **7 **– xxxx - xxxx **8 **– xxxx - xxxx **6 **– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz **9 **– xxxx - xxxx 10– xxxx - xxxx yyyy zzzz yyyy zzzz 11– xxxx - xxxx yyyy zzzz | **7 **– xxxx - xxxx **8 **– xxxx - xxxx **6 **– xxxx - xxxx yyyy zzzz yyyy zzzz yyyy zzzz **9 **– xxxx - xxxx 10– xxxx - xxxx yyyy zzzz yyyy zzzz 11– xxxx - xxxx yyyy zzzz |
| 16·** 15**·** 14**·** 13**·** 12** | 16·** 15**·** 14**·** 13**·** 12** |
| Col1 | Col2 |
|---|---|
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
DOSE 2 puffs | inhaled – four times a day as required 4 more·** paracetamol**·** atenolol**·** ramipril**·** clopidogrel**·** fluticasone**·** tiotropium**·** salbutamol** |
MEDv-183
33
If any of the drug name text (other than the letter ascenders and descenders) is obscured by the boundaries of the list, include that drug in the look-ahead notification
Recommended
Copyright ©2013 Health and Social Care Information Centre
Look-Ahead Scroll Bar
Counts
| Col1 | oxygen 60% – inhalation gas – R continuous – with non re-breather co-amilofruse – 5 mg and 40 mg DOSE 5 mL – oral – once a day salbutamol – 100 micrograms pe inhaler – DOSE 2 puffs – inhaled – | Col3 | Col4 |
|---|---|---|---|
| 4 more ·** paracetamol**·** atenolo** continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day |
| 4 more ·** paracetamol**·** atenolo** continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | 4 more ·** paracetamol**·** atenolo** | ** l** | |
| 4 more ·** paracetamol**·** atenolo** continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | 4 more ·** paracetamol**·** atenolo** |
| Col1 | oxygen 60% – inhalation gas – R continuous – with non re-breather co-amilofruse – 5 mg and 40 mg DOSE 5 mL – oral – once a day salbutamol – 100 micrograms pe inhaler – DOSE 2 puffs – inhaled – | Col3 | Col4 |
|---|---|---|---|
| 4 more ·** paracetamol**·** atenol** continuous – with non re-breath salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | continuous – with non re-breath salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | continuous – with non re-breath salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | continuous – with non re-breath salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day |
| 4 more ·** paracetamol**·** atenol** continuous – with non re-breath salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | 4 more ·** paracetamol**·** atenol** | ** ol** | |
| 4 more ·** paracetamol**·** atenol** continuous – with non re-breath salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | 4 more ·** paracetamol**·** atenol** |
Current Medications
Guidance
MEDv-052
When there are more items than can be displayed in the lookahead notification for current medications, display as many as possible and end the list with a count of the remaining items that could not be displayed
Mandatory
MEDv-053
When a count is displayed in a look-ahead notification and one or more of the medications included in that count have decision support alerts, display a decision support alert icon next to the count
Mandatory
MEDv-055
When displaying current medications only, show drug names and decision support alert icons in the look-ahead notification
Mandatory
| Col1 | oxygen 60% – inhalation gas – R continuous – with non re-breather co-amilofruse – 5 mg and 40 mg DOSE 5 mL – oral – once a day salbutamol – 100 micrograms pe inhaler – DOSE 2 puffs – inhaled – | Col3 | Col4 |
|---|---|---|---|
| 4 more ·** paracetamol**·** atenolo** continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day |
| 4 more ·** paracetamol**·** atenolo** continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | 4 more ·** paracetamol**·** atenolo** | ** l** | |
| 4 more ·** paracetamol**·** atenolo** continuous – with non re-breathe salbutamol– 100 micrograms p inhaler – DOSE 2 puffs– inhaledco-amilofruse– 5 mgand40 mg DOSE 5 mL– oral – once a day | 4 more ·** paracetamol**·** atenolo** |
Copyright ©2013 Health and Social Care Information Centre
34
Guidance
Look-Ahead Scroll Bar
Counts
MEDv-050
is available in a look-ahead notification, display a count instead (as for past medications)
Mandatory
MEDv-051
Do not truncate or abbreviate drug names in the look-ahead notification
Mandatory
| Status | Start Date ▼ | Col3 |
|---|---|---|
| 1 more | ||
| Past Medications | Col2 | Col3 |
|---|---|---|
DOSE 125 micrograms – oral – once a day | DOSE 125 micrograms – oral – once a day | DOSE 125 micrograms – oral – once a day |
hydrochlorothiazide – tablet – DOSE 100 mg – oral – 4 times a daymethyldopa – tablet – DOSE 250 mg – oral – twice a dayfurosemide – tablet – DOSE 40 mg – oral – once a dayDiscontinued Completed Completed | hydrochlorothiazide – tablet – DOSE 100 mg – oral – 4 times a daymethyldopa – tablet – DOSE 250 mg – oral – twice a dayfurosemide – tablet – DOSE 40 mg – oral – once a dayDiscontinued Completed Completed | hydrochlorothiazide – tablet – DOSE 100 mg – oral – 4 times a daymethyldopa – tablet – DOSE 250 mg – oral – twice a dayfurosemide – tablet – DOSE 40 mg – oral – once a dayDiscontinued Completed Completed |
| 3 more | 3 more | |
MEDv-054
counts
35
When displaying past medications only, display counts in the look-ahead notification and not drug names
Mandatory
Copyright ©2013 Health and Social Care Information Centre
Look-Ahead Scroll Bar
Format of Contents
bold
Guidance
MEDv-186
Display drug names in bold and in black text by default
Mandatory
MEDv-187
Display counts and descriptive text (such as ‘more’) in normal weight font
Mandatory
MEDv-188
Use a light solid background colour for the notifications that is both sufficiently different from the colour in the space reserved for notifications and sufficiently different from the black text in the notification
Recommended
MEDv-189
Do not use a border in a dark colour or with a weight greater than 1 point for a look-ahead notification
Recommended
normal weight
| background a light colour | Col2 | Col3 | Col4 |
|---|---|---|---|
| aspirin·** paracetamol** Status Start Date ▼ light colour | aspirin·** paracetamol** Status Start Date ▼ light colour | aspirin·** paracetamol** Status Start Date ▼ light colour | aspirin·** paracetamol** Status Start Date ▼ light colour |
| Status | Status | Start Date ▼ | Start Date ▼ |
| aspirin·** paracetamol** | aspirin·** paracetamol** | aspirin·** paracetamol** | |
Copyright ©2013 Health and Social Care Information Centre
36
Guidance
Look-Ahead Scroll Bar
Format of Contents
delimiter
no delimiter no delimiter
MEDv-056
Use a delimiter that is unlikely to be interpreted as a character or number (such as a black dot ‘�’), with a space either side to separate drug names and to separate the count from drug names
Mandatory
MEDv-184
Do not use leading or trailing delimiters
Mandatory
Copyright ©2013 Health and Social Care Information Centre
| Col1 | Col2 | Col3 | Col4 | Col5 |
|---|---|---|---|---|
| Status | Start Date ▼ | |||
| aspirin·** paracetamol** | aspirin·** paracetamol** | aspirin·** paracetamol** | aspirin·** paracetamol** | |
37
Grouping
Grouping Control
Guidance
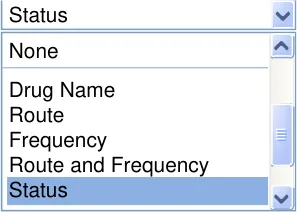
MEDv-084
Provide a standard drop-down list for displaying, selecting and applying grouping to the medications list
Mandatory
MEDv-085
Label the grouping control ‘Group by’
Mandatory
MEDv-190
Include an option in the drop-down list to set the grouping to ‘None’
Mandatory
Copyright ©2013 Health and Social Care Information Centre
38


 Guidance
Guidance
Grouping
Displaying Groups
| Drug Details | Status | Start Date ▼ |
|---|---|---|
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required |
MEDv-083
Present the Medications List View with no grouping active by default
Recommended
| Drug Details | Status | Start Date ▼ |
|---|---|---|
| Inhaled | Inhaled | Inhaled |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required |
| Oral | Oral | Oral |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
MEDv-087
39
Retain the column sort order in the Medications List View when grouping is applied
Mandatory
MEDv-191
Display groups expanded by default
Mandatory
Copyright ©2013 Health and Social Care Information Centre
Grouping
Row Shading
Guidance
MEDv-192
Re-start alternate row shading at the beginning of each group. (Alternate row shading is not needed if there is only one medication in each group)
Mandatory
MEDv-193
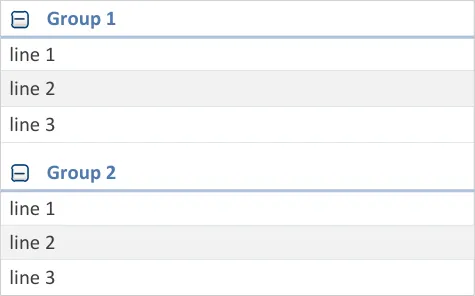
When a group is selected in the grouping control, ensure that at least one group heading is visible in the newly grouped list
Mandatory
Copyright ©2013 Health and Social Care Information Centre
40
 Guidance
Guidance
Grouping
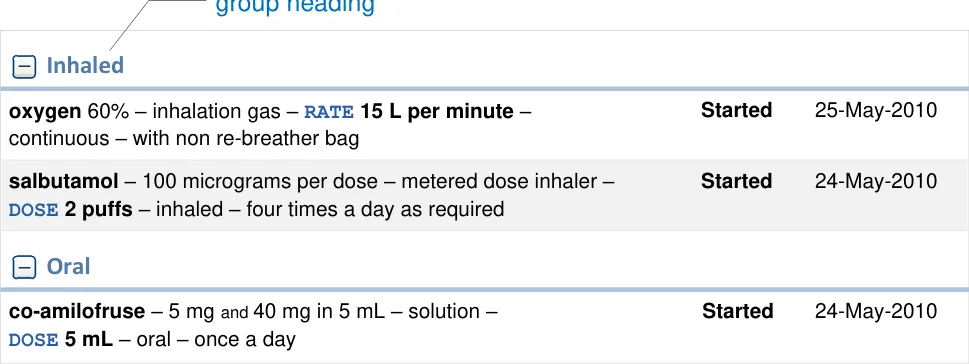
Group Headings
41
MEDv-086
Display clear and prominent headings for each group category
Mandatory
MEDv-090
Do not display group headings for empty groups
Recommended
Copyright ©2013 Health and Social Care Information Centre
Guidance
Grouping
Null Groups
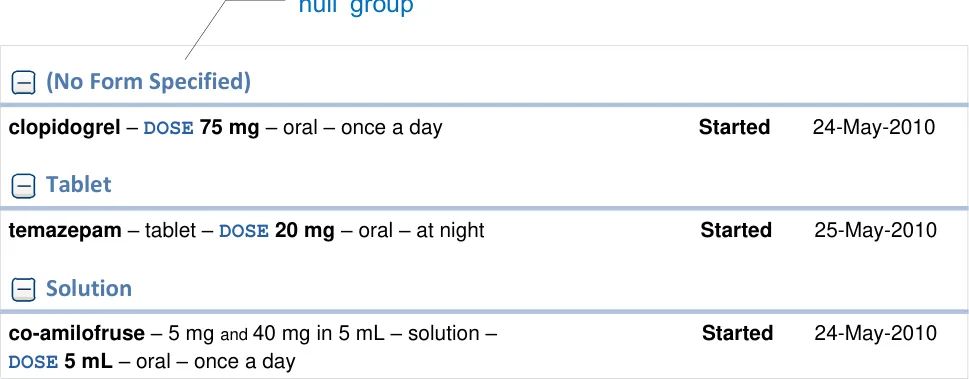
MEDv-091
Provide ‘null’ groups where necessary to support the display of medications that do not have a value for the attribute being used to group the medications
Mandatory
MEDv-194
Display the label for a ‘null’ group heading in brackets
Mandatory
MEDv-195
Display ‘null’ groups at the top of the list of groups
Recommended
Copyright ©2013 Health and Social Care Information Centre
42
Guidance
Grouping
Expand and Collapse


MEDv-092
Provide controls for expanding and collapsing individual groups. Place these controls at the beginning of the group heading
Mandatory
MEDv-089
When a group is collapsed, supplement the group heading with a number representing a count of medications within that group
Mandatory
MEDv-196
Support the selection of group headings and the display of a context menu that includes options for collapsing and expanding all columns
Mandatory
Copyright ©2013 Health and Social Care Information Centre
43
Guidance
Grouping
Combining Group Names
a combined group
| Analgesic (2) | Col2 |
|---|---|
| Analgesic(2) | |
| Analgesic; Non-steroidal Anti-inflammatory(1) | Analgesic; Non-steroidal Anti-inflammatory(1) |
| Non-steroidal Anti-inflammatory(1) | Non-steroidal Anti-inflammatory(1) |
MEDv-198 MEDv-197
Display each medication in only one group (do not duplicate medications so that they can be displayed in more than one group)
MEDv-198
Mandatory
order
MEDv-199
When one or more medications belong to more than one group (such as analgesic and non-steroidal anti-inflammatory), create a new group and label it with the group names combined (such as ‘Analgesic; Non-steroidal Anti-inflammatory’)
Mandatory

When combining group names, display the names in the same order as they would appear in a list that is sorted by that attribute
Mandatory
MEDv-200
When combining group names, separate the labels with a semi-colon
Recommended
Copyright ©2013 Health and Social Care Information Centre
44
Guidance
Sorting
Sorting Control
sort order: most recent first
| Drug Details | Sta | tus | Start Date ▼ |
|---|---|---|---|
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required |
sort order: most recent last
| Drug Details | Sta | tus | Start Date ▲ |
|---|---|---|---|
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
MEDv-101
45
Allow the sort order of a list in the medications list to be changed by clicking on a column heading
Mandatory
MEDv-102
Allow the sort order of a list in the Medications List View to be reversed by clicking on the column heading for the column with the active sort applied
Mandatory
Copyright ©2013 Health and Social Care Information Centre
Guidance
Sorting
Indicating the Sort Order
| sort order symbol column heading | Col2 | Col3 |
|---|---|---|
| Drug Details | Status | Start Date ▼ |
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required |
MEDv-104 MEDv-103
Use an icon or symbol in the column heading to indicate the column by which the data is sorted and the direction of the sort
MEDv-104
Use formatting of the column heading to clearly indicate the column to which the sort order is currently applied
Recommended
Mandatory
MEDv-105
When the sort order is changed from the default to another attribute in the Medications List View, retain the default as a secondary sort order
Mandatory
| Drug Details | Status | Start Date ▼ |
|---|---|---|
| paracetamol Started 26-May-2010 | paracetamol Started 26-May-2010 | paracetamol Started 26-May-2010 |
| oxygen Started 25-May-2010 | oxygen Started 25-May-2010 | oxygen Started 25-May-2010 |
| salbutamol Started 24-May-2010 | salbutamol Started 24-May-2010 | salbutamol Started 24-May-2010 |
| salbutamol Started 12-Apr-2010 | salbutamol Started 12-Apr-2010 | salbutamol Started 12-Apr-2010 |
| paracetamol Started 08-Apr-2010 | paracetamol Started 08-Apr-2010 | paracetamol Started 08-Apr-2010 |
| Drug Details ▼ | Status | Start Date |
|---|---|---|
| oxygen Started 25-May-2010 | oxygen Started 25-May-2010 | oxygen Started 25-May-2010 |
| paracetamol Started 26-May-2010 | paracetamol Started 26-May-2010 | paracetamol Started 26-May-2010 |
| paracetamol Started 08-Apr-2010 | paracetamol Started 08-Apr-2010 | paracetamol Started 08-Apr-2010 |
| salbutamol Started 24-May-2010 | salbutamol Started 24-May-2010 | salbutamol Started 24-May-2010 |
| salbutamol Started 12-Apr-2010 | salbutamol Started 12-Apr-2010 | salbutamol Started 12-Apr-2010 |
Copyright ©2013 Health and Social Care Information Centre
46
Guidance
Levels of Detail
Example 1
+ –
Example 2
Example 3
47
MEDv-201
Provide a control that allows the type and quantity of information displayed to be changed such that the rows and columns may change in number and be presented with a different layout
Recommended
Copyright ©2013 Health and Social Care Information Centre
Guidance
Selection
MEDv-202
Ensure that there are no medications selected by default when a list is opened
Mandatory
| Drug Details | Sta | tus | Start Date ▼ |
|---|---|---|---|
| 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag | 25-May-2010 Started oxygen 60% – inhalation gas – RATE 15 L per minute– continuous – with non re-breather bag |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required |
| Drug Details | Status | Start Date ▼ |
|---|---|---|
oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required |
MEDv-122
Support click (or keyboard selection using the spacebar) to select a medication in the list
Mandatory
Copyright ©2013 Health and Social Care Information Centre
48
Guidance
Selection
Highlighting Selections
MEDv-123
Clearly highlight selected medications in the medication list
Mandatory
| Drug Details | Status | Start Date ▼ |
|---|---|---|
oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required |
| Drug Details | Status | Start Date ▲ |
|---|---|---|
| Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required | Started 24-May-2010 salbutamol– 100 micrograms per dose – metered dose inhaler – DOSE 2 puffs– inhaled – four times a day as required |
co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started | co-amilofruse– 5 mgand40 mg in 5 mL – solution –DOSE 5 mL– oral – once a day24-May-2010 Started |
oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 |
MEDv-124
Maintain the selection of a medication when switching between views of a patient’s medications (such that a medication selected in a Medication List View is automatically selected when switching to the Drug Administration View)
Recommended
MEDv-125
Maintain the selection of a medication when applying or changing a grouping or a sort order and ensure that the selection remains visible
Mandatory
Copyright ©2013 Health and Social Care Information Centre
49
Selection
Supporting Selection
Guidance
MEDv-126
Support the selection of multiple items using CTRL and click for discrete selections, and SHIFT and click for contiguous selections
Mandatory
MEDv-127
Support keyboard-only equivalents such as SHIFT and arrow key for contiguous selection and the CTRL and SPACEBAR to toggle select and deselect when making non-contiguous selections
Mandatory
MEDv-203
When an action is applied to more than one medication, display a summary of the selected medications before allowing the user to complete the action
Mandatory
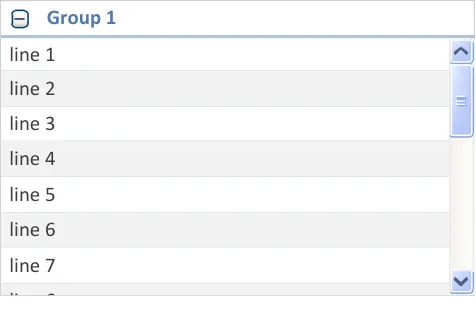
| Drug Details | Status | Start Date ▲ |
|---|---|---|
| line 1 | line 1 | line 1 |
| line 2 | line 2 | line 2 |
| line 3 | line 3 | line 3 |
| line 4 | line 4 | line 4 |
| line 5 | line 5 | line 5 |
| line 6 | line 6 | line 6 |
| line 7 | line 7 | line 7 |
Copyright ©2013 Health and Social Care Information Centre
50
Guidance
Selection
Context Menu
| context menu | Col2 | Col3 | Col4 | Col5 |
|---|---|---|---|---|
| context menu | context menu | context menu | context menu | context menu |
| Drug Details | Drug Details | Status | Status | Start Date ▼ |
oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 Details… | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 Details… | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 Details… | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 Details… | oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 Details… |
oxygen 60% – inhalation gas –RATE 15 L per minute– continuous – with non re-breather bag Started 25-May-2010 Details… | Details… | Details… | Details… | Details… |
co-amilofruse– 5 mgand40 mg in 5 mL – solutioDOSE 5 mL– oral – once a day | n – Suspend | n – Suspend | 24-May-2010 d | 24-May-2010 d |
salbutamol– 100 micrograms per dose – metered dose inhaler –DOSE 2 puffs– inhaled – four times a day as requiredStarted 24-May-2010 Edit | d dose inhaler – Starte Edit | d dose inhaler – Starte Edit | d dose inhaler – Starte Edit | d dose inhaler – Starte Edit |
MEDv-128
Support the display of a context menu for selected medications in the Medications List View (for example, by right-clicking)
Mandatory
MEDv-129
In the context menu, provide appropriate actions and options
Mandatory
51
MEDv-130
In the context menu, support actions with icons where appropriate
Recommended
MEDv-131
In the context menu, grey out actions that are unavailable or disallowed for one or more of the current selections
Mandatory
Copyright ©2013 Health and Social Care Information Centre
Selection
Context Menu
Guidance
MEDv-136
Include an option to access all details for one medication in the context menu
Mandatory
MEDv-132
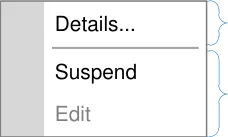
In the context menu, define a consistent and static order of menu items in which frequently used actions are prioritised by placing them higher in the list
Mandatory
MEDv-135
In the context menu for selections in the Medications List View, provide an option for displaying all details for the selected medication
Mandatory
MEDv-133
In the context menu, group similar options so that the direct actions, actions that permit addition of information and actions that display more information, are each grouped together
Mandatory
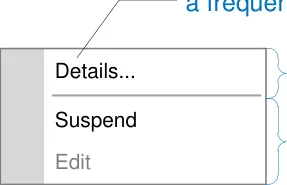
information
actions
Copyright ©2013 Health and Social Care Information Centre
52